Mindray BeneVision N1 Patient Monitor 6660E-PA00017 Accessories Inc! DOM 2021
- Regular price
- $5,995.00
-
$0.00 - Sale price
- $5,995.00
- Unit price
- per
Available in stock: 6
Couldn't load pickup availability
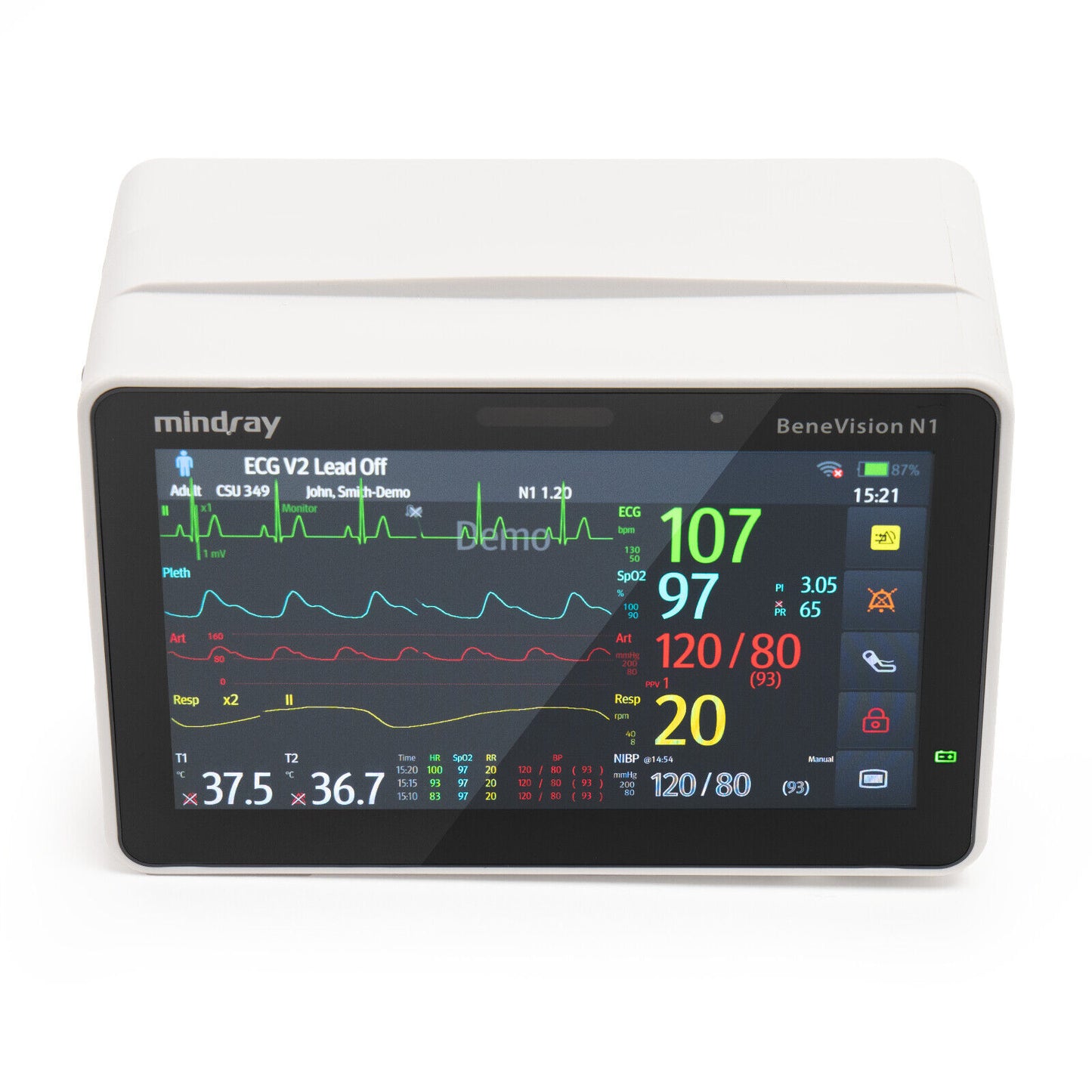
Mindray BeneVision N1 Patient Transport Monitor 6660E-PA00017 DOM: 2021
This Mindray BeneVision N1 Patient Monitor 6660E-PA00017 is in great working and cosmetic condition.
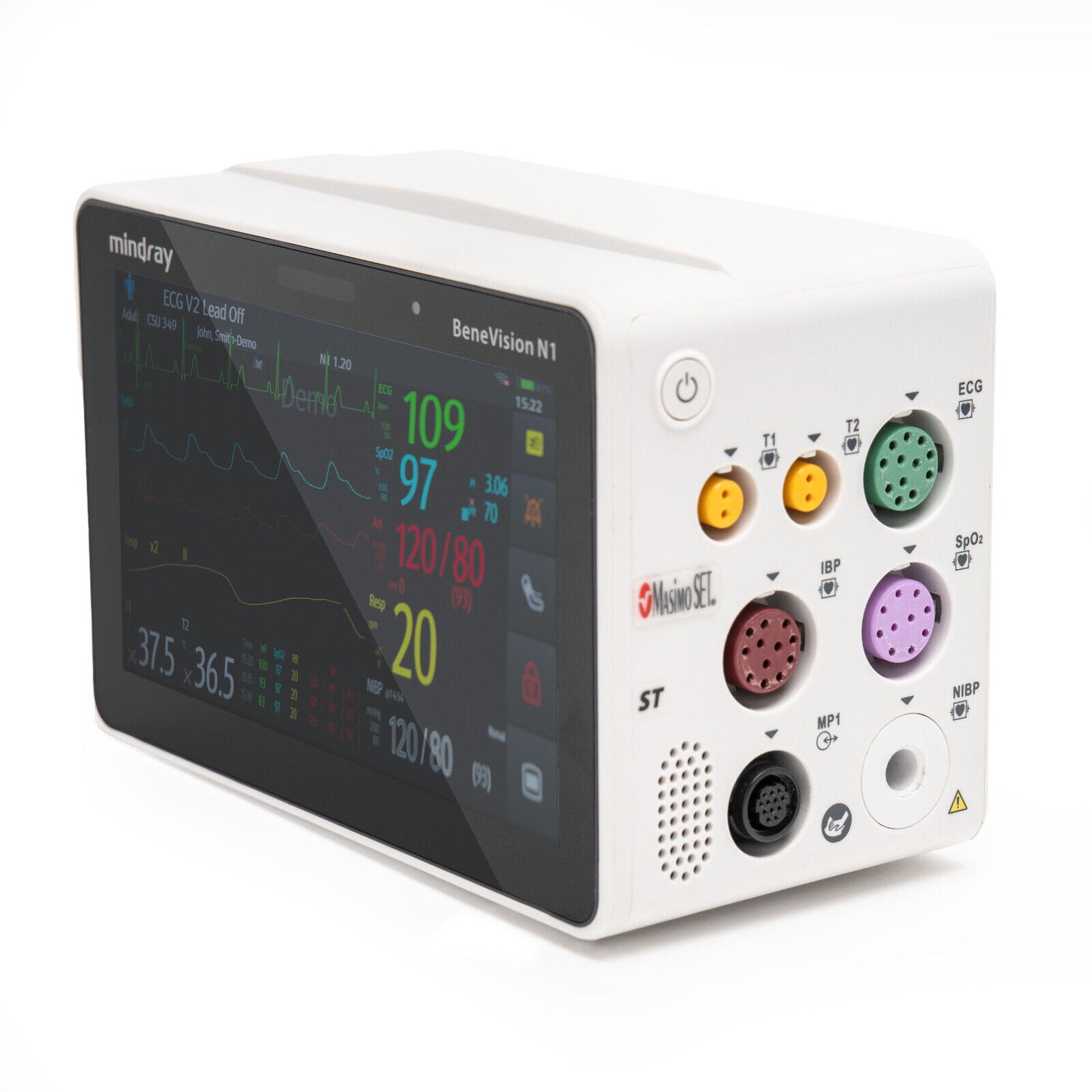
Multipurpose physiological monitor. Features: Defibrillator synchronization, multiple mounting solutions including transport module rack with bedrail hooks. 1.2 m drop test endurance. Data storage up to 48 hours of full disclosure. Masimo SET SpO2, ST/Arrhythmia analysis, 3/5/6/12-lead NIBP, two invasives, dual temperature, integrated battery, respiration, 2.4/5GHz wireless enabled
What's Included:
Mindray BeneVision N1 Patient Monitor; (PN#: 6660E-PA00017); Qty: 1
Adult Accessories - NEW
Mindray Datascope Compatible Reusable Temperature Probe; (REF DMR-AS0); Qty: 1 - NEW
Omni-Kuff Blood Pressure Cuff - Adult Arm Latex-Free; Qty: 1 - NEW
Mindray and Philips Compatible NIBP Hose; (REF AS-16-150); Qty: 1 - NEW
Mindray Datascope Compatible Direct-Connect ECG Cable, (REF C2512P0); Qty: 1 - NEW
___________________
This device has been carefully reviewed and conforms with eBay's Prescription devices policy (https://www.ebay.com/help/policies/prohibited-restricted-items/medicine-medical-devices-healthcare-products-policy?id=4322) .
To the best of our knowledge and abilities, this device has been cleaned and handled in accordance with manufacturer's instructions.
"The sale of this item may be subject to regulation by the U.S. Food and Drug Administration and state and local regulatory agencies. If so, do not bid on this item unless you are an authorized purchaser. If the item is subject to FDA regulation, I will verify your status as an authorized purchaser of this item before shipping of the item."
---
Also per eBay's Prescription Devices Policy, we must provide the following information about our company for listings of devices that may fall under this FDA guideline:
KenMed Supply, LLC, d.b.a, KenMed Surgical
Casselberry, Florida
[phone removed by eBay] - Office Number